|
CEE 680 |
|
18 December 2006 |
FINAL EXAM
Closed book, three pages of notes allowed.
Answer Question A and either B or C. Please state any additional assumptions you made, and show all work. If you don’t have time to complete a section, please describe how you would solve the problem (without using a computer program such as MINEQL).
Part I: Answer Question A
A. Solubility & Predominance. (75%)
Gypsum (CaSO4.2H2O (s)) and Calcium Hydroxide (Ca(OH)2 (s)) are two important calcium-bearing minerals.
1. Prepare a solubility diagram (log C vs pH) for a water in equilibrium with calcium hydroxide and Gypsum. Assume the water has 10-2 M total sulfates. Show all soluble species along with the CaT line and indicate where precipitation will occur and the type of precipitate. (35%)
2. Prepare a predominance diagram, showing the precipitates and major soluble species (in areas where there are no precipitates). As would be typical for a problem of this type, make pH the x-axis, and log total sulfate (SO4T), the y-axis. Assume a total soluble calcium concentration of 10 mM.. Note that this is a very simple system with only 2 soluble species. (35%)
3. Describe in a qualitative way how the Calcium solubility would change if we added carbonate species. (5%)
Answer to A.1.
a. Using the equilibria
for calcium hydroxide from below:
![]()
we get the following for the free aquo ion:
![]()
![]()
![]()
and for the monohydroxide:
![]()
![]()
![]()
![]()
![]()
![]()
b. Now moving on to
the calcium sulfate precipitate
![]()
for free aquo calcium ion we get:
![]()
![]()
For pHs below 2, the amphoteric form predominates (HSO4-) and
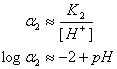
And with an ST of 10-2M, the above equation becomes:
![]()
![]()
and for pHs above 2 where essentially all of the soluble sulfate species are the fully deprotonated anion (SO4-2), we can equate total sulfates to the SO4-2 species:
![]()
![]()
and for the monohydroxide (CaOH+):
![]()
![]()
![]()
For pHs below 2, the amphoteric form predominates (HSO4-) and with an ST of 10-2M, the above equation becomes:
![]()
and for pHs above 2 where essentially all of the soluble sulfate species are the fully deprotonated anion (SO4-2), we can equate total sulfates to the SO4-2 species:
![]()
![]()
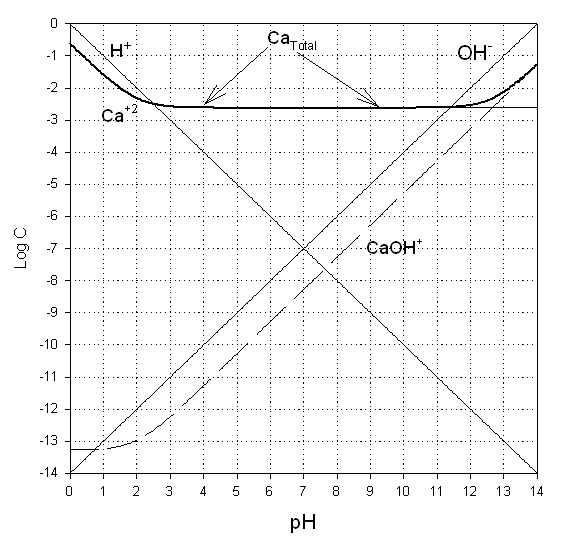
Now combining, we find that the hydroxide controls at pHs of 12.7 and above. Below this level, the gypsum is less soluble.
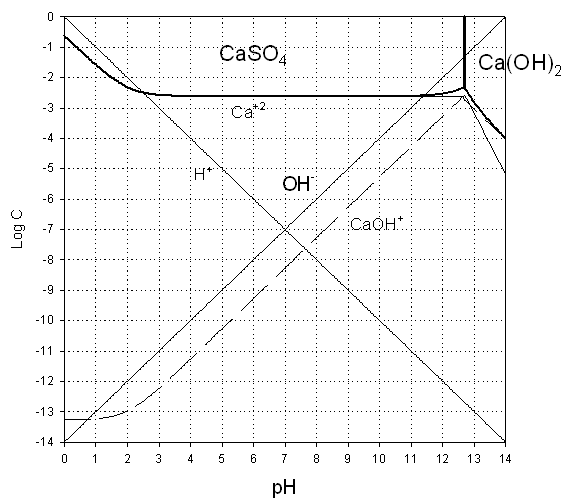
Answer to A.2.
Type A lines
First, lets consider the boundary between the aquo ion and the monohydroxide
![]()
When they’re present at equal concentrations:
![]()
or:
![]()
so:
pH = 12.85
Type B line
Next, we will want to look at the Ca+2/Ca(OH)2 boundary
![]()
now adopting a total Ca of 10 mM, we get:
![]()
or:
pH =12.405
Next, lets look at the Ca+2/CaSO4 boundary:
![]()
now adopting a total Ca of 10 mM, we get:
![]()
![]()
logST = -2.62 –logα2
For pHs below 2, the amphoteric form predominates and
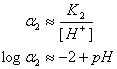
So the equation becomes:
logST = -2.62 –(-2+pH)
logST = -0.62-pH
and for pHs above 2 where essentially all of the soluble sulfate species are the fully deprotonated anion, we can equate total sulfates to the SO4-2 species:
logST = -2.62
Type C line
Finally, lets consider the boundary between the two solid phases. Predominance regions appear to switch just about the point where calcium aquo ion and the monohydroxide are equal, so lets take the aquo ion equations.
First, from the hydroxide solid solubility, we have:
![]()
And from the gypsum solubility we have:
![]()
Combining, we get:
![]()
![]()
And since the two phases only coexist at higher pHs, we only need to consider the case where total sulfate equals the completely deprotonated sulfate anion.
![]()
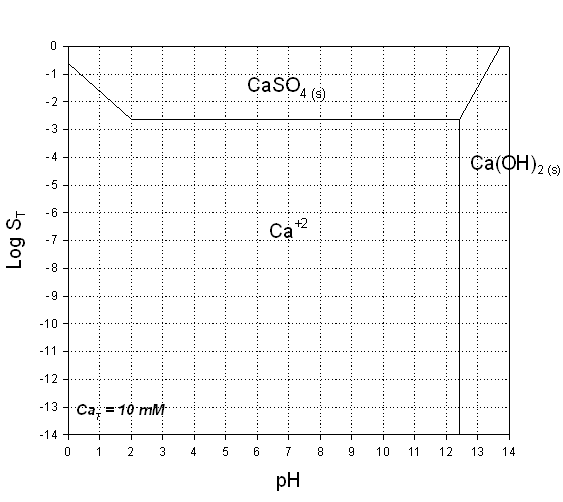
Answer to A.3.
If enough carbonate was present, the calcium solubility would be depressed, especially at high pHs. There would also be a calcium carbonate phase zone in predominance diagrams.
Part II:. Answer either B or C
B. Redox (25%)
Monochloramine (NH2Cl) is a widely used disinfectant for drinking waters and wastewaters. Its final reduced form is usually chloride and ammonia. While monochloramine is both a disinfectant and an oxidant, it is not a very powerful one. You have been asked to comment on the possibility that application of monochloramine to a water containing naturally-occurring bromide could oxidize that anionic species to hypobromous acid..
1. Write a balanced equation for the oxidation of bromide to hypobromous acid by monochloramine (10%)
2. Determine the stoichiometry of this reaction (e.g., mg-NH2Cl/mg-Br). (5%)
3. Determine the log K for this reaction (5%)
4. Calculate the equilibrium ratio of HOBr to bromide at pH 7, and in the presence of 10-4 M monochloramine, 10-4.5 M total free ammonia, and 10-3 M chloride. (5%)
Answer to B.1.
|
Equ# |
Half
Cell Reaction |
DEo (Volts) |
|
-7 |
½Br- + ½H2O =
½HOBr + ½H+ + e- |
-1.33 |
|
11 |
½NH2Cl + H+ +e-
= ½Cl- + ½NH4+ |
+1.40 |
|
|
½Br- + ½NH2Cl + ½H+
+ ½H2O = ½HOBr + ½Cl- + ½NH4+ |
+0.07 |
Or simplifying by making it a 2 electron transfer reaction:
Br-
+ NH2Cl + H+ + H2O = HOBr + Cl- +
NH4+
Answer to B.2.
Stoichiometry:
![]()
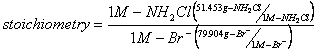
![]()
Answer to B.3.
Let’s consider the following reaction:
Br- +
NH2Cl + H+ + H2O = HOBr + Cl- + NH4+
![]()
Expressed this way, it is a 2-electron transfer
![]()
![]()
Answer to B.4.
For the reaction:
Br- +
NH2Cl + H+ + H2O = HOBr + Cl- + NH4+
![]()
![]()
under the cited conditions:
![]()
![]()
C. Metal Matching (25%)
Write the letter of the correct answer in the box at left. Each answer is used only once[1].
|
Correct Answer |
Questions |
|
Possible Answers |
|
I |
1. This metal has a pKso with carbonate of 8.34 |
6 |
A. Iron |
|
E |
2. The pe0 for the reduction of the divalent form of this metal to its zero valent form is about -12.9 |
7 |
B. Copper |
|
G |
3. The pe0 for the reduction of the divalent form of this metal to its zero valent form is about -4.0 |
9 |
C. Arsenic |
|
H |
4. This divalent metal has an α0 of about 0.92 at pH 9 |
5 |
D. Lead |
|
D |
5. This divalent metal has an α0 of about 0.05 at pH 9 |
2 |
E. Zinc |
|
A |
6. This divalent metal has an α0 of about 0.76 at pH 9 |
10 |
F. Aluminum |
|
B |
7. This metal has the highest ligand affinity of all in the Irving-Williams series |
3 |
G. Nickel |
|
J |
8. This metal does not form complexes with EDTA |
4 |
H. Cadmium |
|
C |
9. This metal forms oxo ions that are predomant under natural aquatic conditions |
1 |
I. Calcium |
|
F |
10. This metal forms strong complexes with fluoride, but does not normally undergo oxidation or reduction reactions in solution |
8 |
J. None of the above |
Best to calculate the pe0 and α values from the thermodynamic data for the metals listed:
![]()
Some important equilibrium constants:
|
Equilibria |
Log K |
α0 at pH 9 |
|
Mg(OH)2 (s) = Mg+2 + 2OH- |
-11.6 |
|
|
Fe+3 + H2O = FeOH+2 + H+ |
-2.19 |
|
|
Mg+2 + H2O = MgOH+ + H+ |
-11.44 |
0.9964 |
|
MgCO3 (s) = Mg+2 + CO3-2 |
-7.5 |
|
|
CaCO3(s) = Ca+2 + CO3-2 |
-8.34 |
|
|
Ca(OH2)(s) = Ca+2 + 2OH- |
-5.19 |
|
|
CaSO4.2H2O(s)
= Ca+2 + SO4-2 + 2H2O |
-4.62 |
|
|
CaOH+ = Ca+2 + |
-1.15 |
0.9999 |
|
AlOH+2 = Al+3 + |
-9.01 |
0.0001 |
|
CdOH+ = Cd+2 + |
-3.92 |
0.9232 |
|
Co |
-4.80 |
0.6131 |
|
Cu |
-6.00 |
0.0909 |
|
FeOH+ = Fe+2 + |
-4.50 |
0.7597 |
|
Hg |
-10.60 |
0.0000 |
|
NiOH+ = Ni+2 + |
-4.14 |
0.8787 |
|
Pb |
-6.29 |
0.0488 |
|
Zn |
-5.04 |
0.4770 |
Some
important half-cell reactions
|
Equ# |
Half
Cell Reaction |
DEo (Volts) |
Log K |
pe |
|
1 |
O2(g) + 4H+ + 4e- = 2H2O |
+1.23 |
|
|
|
2 |
Mn+3 +
e- = Mn+2 |
+1.51 |
|
|
|
3 |
Mn+4 +
e- = Mn+3 |
+1.65 |
|
|
|
4 |
MnO4- + 8H+ + 5e- = Mn+2 + 4H2O |
+1.49 |
|
|
|
5 |
Fe+3 +
e- = Fe+2 |
+0.77 |
|
|
|
6 |
Cu+2 + e- = Cu+ |
+0.16 |
|
|
|
7 |
½HOBr + ½H+ + e- =
½Br- + ½H2O |
+1.33 |
|
|
|
8 |
O3 (g) + 2H+ + 2 e- = O2 (g) + H2O |
+2.07 |
|
|
|
9 |
Al+3 + 3e- = Al(s) |
-1.68 |
|
|
|
10 |
S(s) + 2H+ + 2e- = H2S (g) |
+0.17 |
|
|
|
11 |
½NH2Cl + H+ +e-
= ½Cl- + ½NH4+ |
+1.40 |
|
|
|
12 |
Zn+2 +
2e- = Zn(s) |
-0.76 |
-25.76 |
-12.88 |
|
13 |
Ni+2 +
2e- = Ni(s) |
-0.24 |
-7.98 |
-3.99 |
|
14 |
Pb+2 +
2e- = Pb(s) |
-0.13 |
-4.27 |
-2.14 |
|
15 |
Cu+2 +
2e- = Cu(s) |
+0.34 |
11.48 |
5.74 |
|
16 |
Hg2+2 +
2e- = 2Hg(l) |
+0.91 |
30.79 |
15.40 |
|
17 |
Fe+2 +
2e- = Fe(s) |
-0.44 |
-14.9 |
-7.45 |
Properties of Selected Elements
|
Element |
Symbol |
Atomic # |
Atomic Wt. |
|
Electronegativity |
|
|
Aluminum |
Al |
13 |
26.98 |
3 |
1.47 |
|
|
Bromine |
Br |
35 |
79.904 |
1,3,5,7 |
2.74 |
|
|
Calcium |
Ca |
20 |
40.08 |
2 |
1.04 |
|
|
Carbon |
C |
6 |
12.01 |
2,4 |
2.50 |
|
|
Chlorine |
Cl |
17 |
35.453 |
1,3,5,7 |
2.83 |
|
|
Copper |
Cu |
29 |
63.54 |
1,2 |
1.75 |
|
|
Hydrogen |
H |
1 |
1.01 |
1 |
2.20 |
|
|
Magnesium |
Mg |
12 |
24.31 |
2 |
1.23 |
|
|
Manganese |
Mn |
25 |
54.94 |
2,3,4,6,7 |
1.60 |
|
|
Nitrogen |
N |
7 |
14.0047 |
3,5 |
3.07 |
|
|
Oxygen |
O |
8 |
16.00 |
2 |
3.50 |
|
|
Potassium |
K |
19 |
39.10 |
1 |
0.91 |
|
|
Sodium |
Na |
11 |
22.99 |
1 |
1.01 |
|
|
Strontium |
Sr |
38 |
87.62 |
2 |
0.99 |
|
|
Sulfur |
S |
16 |
32.06 |
2,4,6 |
2.44 |
|
Selected Acidity Constants
(Aqueous Solution,
25°C, I = 0)
|
NAME |
FORMULA |
pKa |
|
Perchloric acid |
HClO4 = H+ + ClO4- |
-7 |
|
Hydrochloric acid |
HCl = H+ + Cl- |
-3 |
|
Sulfuric acid |
H2SO4= H+ + HSO4- |
-3 |
|
Nitric acid |
HNO3 = H+ + NO3- |
0 |
|
Bisulfate ion |
HSO4- = H+ +
SO4-2 |
2 |
|
Phosphoric acid |
H3PO4 = H+ + H2PO4- |
2.15 |
|
o-Phthalic acid |
C6H4(COOH)2 = H+ + C6H4(COOH)COO- |
2.89 |
|
p-Hydroxybenzoic acid |
C6H4(OH)COOH = H+ + C6H4(OH)COO- |
4.48 |
|
Nitrous acid |
HNO2 = H+ + NO2- |
4.5 |
|
Acetic acid |
CH3COOH = H+ + CH3COO- |
4.75 |
|
Aluminum ion |
Al(H2O)6+3
= H+ + Al(OH)(H2O)5+2 |
4.8 |
|
Carbonic acid |
H2CO3 = H+ + HCO3- |
6.35 |
|
Hydrogen sulfide |
H2S = H+ + HS- |
7.02 |
|
Dihydrogen phosphate |
H2PO4- = H+ + HPO4-2 |
7.2 |
|
Hypochlorous acid |
HOCl = H+
+ OCl- |
7.5 |
|
Hypobromous acid |
HOBr = H+
+ OBr- |
8.71 |
|
Ammonium ion |
NH4+ = H+ + NH3 |
9.24 |
|
Bicarbonate ion |
HCO3- = H+ + CO3-2 |
10.33 |
|
Monohydrogen phosphate |
HPO4-2 = H+ + PO4-3 |
12.3 |