|
CEE 577 |
8 April 2002 |
MID-TERM EXAM
Closed
book, 1 sheet of notes allowed.
Answer
both questions. Please state any
additional assumptions you made, and show all work.
I.
(50%)
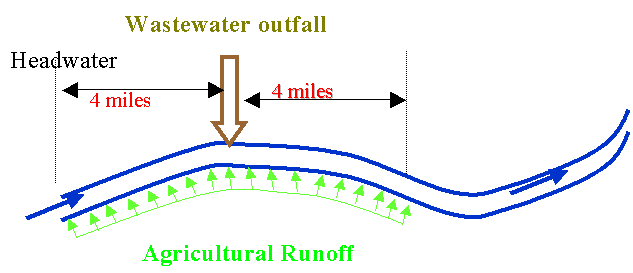
Bovine Brook emerges from pristine headwaters and runs through an agricultural region
before it reaches the city of Poultryville.
The BOD of the headwaters is 4 mg/L, and the dissolved oxygen is 8.8
mg/L (saturation is 10.1 mg/L for 15oC). Starting at mile point zero, there is a significant non-point
agricultural runoff of BOD amounting to 40 kg/mile/day. At mile point 4.0, the stream is met by the
Poultryville WWTP outfall. Here a WW
flow of 1 cfs is discharged with a CBOD of 150 mg/L, an ammonia-N concentration
of 24 mg-N/L and a DO of 6 mg/L.
Immediately past this outfall is 4 more miles of agricultural land. This is better managed than the upstream
land, so that the runoff BOD averages 15 kg/mile/day. Calculate the dissolved oxygen concentration 4 miles
downstream of the Poultryville WWTP outfall (T=15oC). Assume the flow is constant at 40 cfs from
the headwaters to the end of the non-point agricultural runoff. You may also assume an SOD downstream of the
WWTP outfall of 2.0 g/m2/d.
Additional Information:
U = 0.150 ft/sec = 2.45 miles/day T
= 15oC
DOsat or Cs = 10.1 mg/L (at 15oC) H
= 4 ft = 1.22 m
BOD deoxygenation rate (kN = kd)
= 0.8 day-1 (at 15oC) for kN and kd,
=1.047
CBOD settling rate (ks) = 0.080 day-1 (at 15oC) for reareation, =1.024
SOLUTION:
Calculate ka and adjust for temperature:
ka = 0.555 d-1
Divide stream into two reaches of 4 miles in length. The travel time for each is:
t = 1.63 d
The non-point CBOD load is:
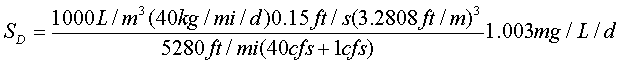
Then determine the BOD and deficit at the end of the first reach:
Overall deficit = terms incorporating (CBOD point deficit + point BOD + distributed deficit + distributed BOD)
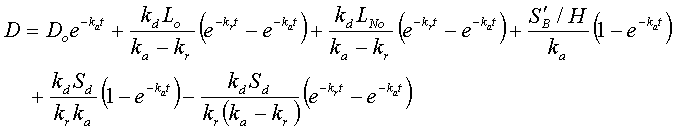
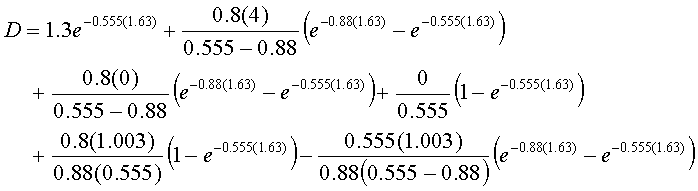
D = 0.526 + 1.639 + 0 + 0 + 0.978 – 0.467
D = 2.677
C = 10.1 – 2.677 = 7.423 mg/L
Calculate the BOD and deficit at the junction of the two reachs and the point source:
Finally, determine the BOD and deficit at the end of the second reach:
The non-point CBOD load is:
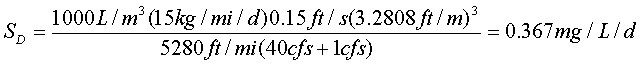
Overall deficit = terms incorporating (CBOD point deficit + point BOD + distributed deficit + distributed BOD)
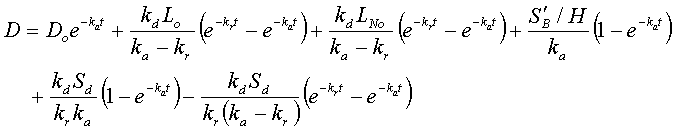
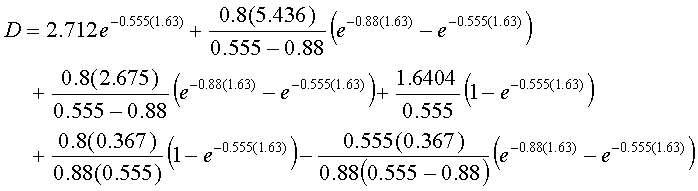
D = 1.10 + 2.23 + 1.16 + 1.76 + 0.36 – 0.17
D = 6.44
C = 10.1 – 6.44 = 3.66 mg/L
