|
CEE 577 |
8 April 2002 |
MID-TERM EXAM
Closed
book, 1 sheet of notes allowed.
Answer
2 of the following 3 questions. Please
state any additional assumptions you made, and show all work.
I.
(50%)
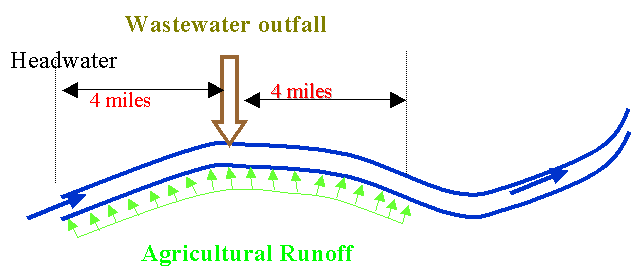
Bovine Brook emerges from pristine headwaters and runs through an agricultural
region before it reaches the city of Poultryville. The BOD of the headwaters is 4 mg/L, and the dissolved oxygen is
8.8 mg/L (saturation is 10.1 mg/L for 15oC). Starting at mile point zero, there is a
significant non-point agricultural runoff of BOD amounting to 40
kg/mile/day. At mile point 4.0, the
stream is met by the Poultryville WWTP outfall. Here a WW flow of 1 cfs is discharged with a CBOD of 150 mg/L, an
ammonia-N concentration of 24 mg-N/L and a DO of 6 mg/L. Immediately past this outfall is 4 more
miles of agricultural land. This is
better managed than the upstream land, so that the runoff BOD averages 15
kg/mile/day. Calculate the
dissolved oxygen concentration 4 miles downstream of the Poultryville WWTP
outfall (T=15oC). Assume
the flow is constant at 40 cfs from the headwaters to the end of the non-point
agricultural runoff. You may also
assume an SOD downstream of the WWTP outfall of 2.0 g/m2/d.
Additional Information:
U = 0.150 ft/sec = 2.45 miles/day T
= 15oC
DOsat or Cs = 10.1 mg/L (at 15oC) H
= 4 ft = 1.22 m
BOD deoxygenation rate (kN = kd)
= 0.8 day-1 (at 15oC) for kN and kd,
=1.047
CBOD settling rate (ks) = 0.080 day-1 (at 15oC) for reareation, =1.024
II.
(50%) Silver lake is an urban surface water that
has received inputs of nickel for many years.
Efforts to clean up the lake have resulted in the termination of all
nickel loads. Lake levels have dropped
due to decay (0.00005 d-1) and hydraulic flushing. Unfortunately, Senator Porkbarrel has
succeeded in getting his brother's chrome fender business exempted from the
nickel ban. The amount on nickel
discharged is directly related to the amount of product produced. Due to changes in the automotive industry,
production has dropped off since the business began (see table below). During this time nickel waste has been
constant at 3.3 Kg Ni per ton of product.
The output data below were collected for a few selected years. On January 1, 1985, the lake concentration
of nickel was 0.13 mg/L. The lake has a
volume of 12.5 million cubic meters, a surface area of 3 million square meters,
and an outflow of 8000 cubic meters per day.
Ignore any temperature effects.
Porkbarrel Plating Co. Data
|
Year |
Output (tons/yr) |
|
1965 |
580 |
|
1975 |
510 |
|
1985 |
440 |
|
1995 |
370 |
- Calculate the expected
nickel concentration at the beginning of the year 2010.
- Determine the year when
the maximum Ni concentration occurred or will occur.
III. (50%) On a separate sheet of paper, answer
any five (5) of the following questions.
A.
Describe
the difference between mechanistic and empirical modeling
B.
Is
a first order reaction always faster than a zero order reaction? Explain.
C.
Describe
the differences between point and non-point loading and give examples.
D.
Explain
the relationship between Secchi-disk depth and lake trophic state. Why is there such a relationship?
E.
Describe
3 different methods for determining stream velocity.
F.
Explain
what the light and dark bottle method measures and how it works
G.
Describe
the factors that determine re-aeration in rivers, and contrast this with the
factors that determine re-aeration in lakes.
In your description, relate micro-scale processes (molecules) to
macro-scale (bulk water or air)