|
CEE 370 |
Fall 2006 |
Exam #1
October 16, 2006
Closed Book, one sheet of notes allowed
Please answer any 4 of the following 9 questions. Each is worth 25 points. Show all work. Be neat, and box-in your answer.
1. Calculate the ThOD (in mg/L) of the following solutions: (25 points)
a. 10-3 moles/L of sodium nitrite, NaNO2
The balanced stoichiometric equation is:
![]()
The ThOD is then:
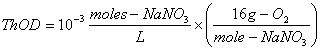
![]()
b. 3x10-3 moles/L of formic acid, HCOOH
The balanced stoichiometric equation is:
![]()
The ThOD is then:
![]()
![]()
c. 10-2 moles/L of ammonium carbonate, (NH4)2CO3
The balanced stoichiometric equation is:
![]()
The ThOD is then:
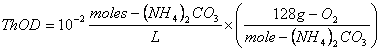
![]()
2. Perform a charge balance on the following water. As part of this balance determine the amount of unbalanced charge and its percentage of the total. What does this number tell you about the accuracy of the chemical analysis? (25 points)
|
Chemical Substance |
Conc. (mg/L) |
GFW |
Conc.
(mM) |
Conc.
(meq/L) |
|
Na+ |
12.5 |
22.9898 |
0.5437 |
0.5437 |
|
K+ |
1.1 |
39.0983 |
0.0281 |
0.0281 |
|
Ca+2 |
39.3 |
40.0800 |
0.9805 |
1.9611 |
|
Mg+2 |
3.5 |
24.3050 |
0.1440 |
0.2880 |
|
NO3- |
8.8 |
62.0049 |
0.1419 |
0.1419 |
|
SO4-2 |
12.4 |
96.0576 |
0.1291 |
0.2582 |
|
Cl- |
33.9 |
35.4530 |
0.9562 |
0.9562 |
|
HCO3- |
112.3 |
61.0171 |
1.8405 |
1.8405 |

3. What is the TDS (in mg/L), and the ionic strength for the water in question #2? (25 points)
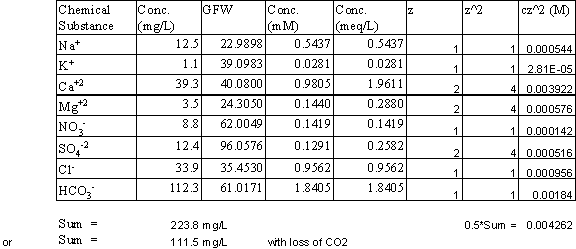
TDS = 224 or 112 mg/L
Ionic Strength = 4.3 mM
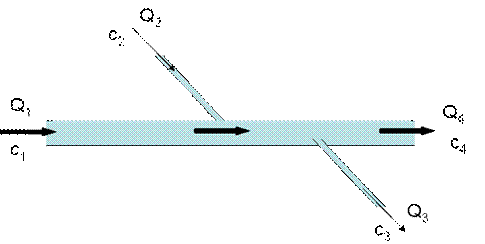
4. The diagram below shows a long deep-rock tunnel carrying water. There is one major inflow (Q1), one minor inflow (Q2), one minor outflow (Q3) and the major outflow (Q4), all but one of which are known. In addition, the concentration of chloride (a conservative substance) is known at 2 of the 4 inflow/outflow locations. Calculate the missing flow (Q3) and the missing chloride concentrations (c3 and c4) for this system (25 points)
|
Location |
Q (cfs) |
Chloride (mg/L) |
|
1 (main inflow) |
15 |
2 |
|
2 (side inflow) |
3 |
6 |
|
3 (side outflow) |
??? |
??? |
|
4 (main outflow) |
17 |
??? |
Mass Balance on Water across the entire system

Mass Balance on chloride for the system:

Recognize that we’re dealing with a conservative substance. This means that the chloride doesn’t decay. For this reason, the concentration will be constant downstream of the last input. Thus, c3 = c4.
![]()
5. Use the following data to estimate the rate constant for the loss of chlorine dioxide in water. Assume the reaction is first order in chlorine dioxide (25 points)
|
Time (min) |
Concentration (µM) |
k (min-1) |
|
|
ti-1 to ti |
t0 to ti |
||
|
0 |
100 |
|
|
|
50 |
22 |
0.03028 |
|
|
100 |
5 |
0.02996 |
0.02963 |
|
Average: |
0.03012 |
|
|
![]()
or
![]()
So:

And we can do this several ways, as there are three sets of paired C-t data. While there is some varability among these it is rather small, and the rate constant can be said to be about
k = 0.030 min-1 = 5x10-4 sec-1
6. True/False. Indicate whether the following statements are true (T) or false (F). (25 points total; 2 points each; 1 free point)
|
1. |
T |
The
Arrhenius equation allows one to adjust rate constants from one temperature
to another |
|
2. |
F |
A triprotic
acid has three times the strength as a monoprotic acid |
|
3. |
T |
A conjugate
base is what forms when an acid losses a proton |
|
4. |
T |
When an
organic compound name ends in “ol”, it usually means the compound is an
alcohol |
|
5. |
T |
Catalysts
accelerate chemical reactions by lowering the activation energy |
|
6. |
F |
When Gibbs
Free Energy increases, the reaction will tend to go forward |
|
7. |
T |
Alkynes all
have triple bonds |
|
8. |
T |
Henry’s law
describes the relationship between partial pressure and dissolved
concentration |
|
9. |
F |
BTEX is a
new type of dog food |
|
10. |
T |
Changes in
ionic strength can cause shifts in chemical equilibria |
|
11 |
F |
An element
of high electronegativity will share its bonding electrons equally with an
element of low electronegativity |
|
12 |
F |
Biochemical
oxygen demand (BOD) differs from theoretical oxygen demand (ThOD) in that BOD
only refers to oxygen demand from pharmaceuticals |
7. The half life for nonylphenol in groundwater is estimated to be 35 days. If the concentration of nonylphenol in a groundwater plume is currently 12 ppb, how long will it take for the concentration to drop below 1 ppb? (25 points)
Since I give you a half-life, its reasonable to assume this is a first order process, so
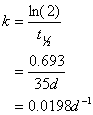
And for 1st order reactions:
![]()
Which gives us:

≈125 days
8. The equilibrium constant for the following reaction is 10-10.3. What percent of the total carbonate is in the form of HCO3- at pH 11.5? (25 points)
HCO3- = CO3-2 + H+
The equilibrium expression for this reaction is:
![]()
So:
![]()
And therefore:
![]()
Thus:
![]()
And recognizing that at such a high pH, virtually all of the carbonate are in the form of bicarbonate and carbonate (i.e., there is essetially no carbonic acid):
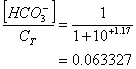
Or HCO3-/total carbonate ≈ 6.3%
9. You need to oxidize butyric acid (CH3CH2CH2COOH) completely to carbon dioxide (CO2) by addition of potassium dichromate (K2Cr2O7). After the reaction is complete, the chromium is in the trivalent form (Cr+3). How many mg/L of potassium dichromate are needed to completely oxidize 4.6 mg/L of butyric acid? (25 points).
This is a redox (reduction-oxidation) reaction, so the electron transfer must be balanced. This requires that we first separate out the two half reactions,
First the oxidation half reaction we have carbon becoming oxidized:
![]()
Next, dichromate must be the substance that is reduced (i.e., it does the oxidizing of the butyric acid). We’re also told that an end product is Cr+3. So the reduction half reaction is:
![]()
Then combine to balance the overall equation:
3 x (![]() )
)
10 x (![]() )
)
![]()
Or
![]()
Next calculate the mass requirements
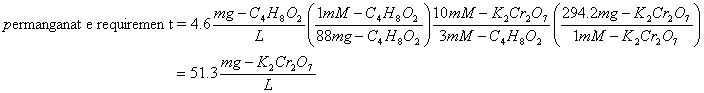
Appendix
Selected Chemical Constants
|
Element |
Symbol |
Atomic # |
Atomic Wt. |
|
Electronegativity |
|
|
Aluminum |
Al |
13 |
26.98 |
3 |
1.47 |
|
|
Boron |
B |
5 |
10.81 |
3 |
2.01 |
|
|
Calcium |
Ca |
20 |
40.08 |
2 |
1.04 |
|
|
Carbon |
C |
6 |
12.01 |
2,4 |
2.50 |
|
|
Chlorine |
Cl |
17 |
35.453 |
1,3,5,7 |
2.83 |
|
|
Chromium |
Cr |
24 |
52.00 |
many |
1.56 |
|
|
Helium |
He |
2 |
4.00 |
0 |
|
|
|
Holmiuum |
Ho |
67 |
164.93 |
3 |
1.10 |
|
|
Hydrogen |
H |
1 |
1.01 |
1 |
2.20 |
|
|
Magnesium |
Mg |
12 |
24.31 |
2 |
1.23 |
|
|
Manganese |
Mn |
25 |
54.94 |
2,3,4,6,7 |
1.60 |
|
|
Nitrogen |
N |
7 |
14.01 |
many |
3.07 |
|
|
Oxygen |
O |
8 |
16.00 |
2 |
3.50 |
|
|
Potassium |
K |
19 |
39.10 |
1 |
0.91 |
|
|
Sodium |
Na |
11 |
22.99 |
1 |
1.01 |
|
|
Sulfur |
S |
16 |
32.06 |
2,4,6 |
2.44 |
|
Selected Acidity Constants (Aqueous Solution, 25°C, I = 0)
|
NAME |
FORMULA |
pKa |
|
|
Hydrochloric acid |
HCl = H+ + Cl- |
-3 |
|
|
Sulfuric acid |
H2SO4=
H+ + HSO4- |
-3 |
|
|
Nitric acid |
HNO3 = H+ +
NO3- |
-0 |
|
|
Bisulfate ion |
HSO4-
= H+ + SO4-2 |
2 |
|
|
Phosphoric acid |
H3PO4 =
H+ + H2PO4- |
2.15 |
|
|
Hydrofluoric acid |
HF = H+ + F- |
3.2 |
|
|
Nitrous acid |
HNO2 = H+ + NO2- |
4.5 |
|
|
Acetic acid |
CH3COOH = H+ + CH3COO- |
4.75 |
|
|
Propionic acid |
C2H5COOH
= H+ + C2H5COO- |
4.87 |
|
|
Carbonic acid |
H2CO3 =
H+ + HCO3- |
6.35 |
|
|
Hydrogen sulfide |
H2S = H+ + HS- |
7.02 |
|
|
Dihydrogen phosphate |
H2PO4-
= H+ + HPO4-2 |
7.2 |
|
|
Hypochlorous acid |
HOCl = H+ + OCl- |
7.5 |
|
|
Ammonium ion |
NH4+
= H+ + NH3 |
9.24 |
|
|
Hydrocyanic acid |
HCN = H+ + CN- |
9.3 |
|
|
Phenol |
C6H5OH
= H+ + C6H5O- |
9.9 |
|
|
Bicarbonate ion |
HCO3-
= H+ + CO3-2 |
10.33 |
|
|
Monohydrogen phosphate |
HPO4-2 = H+ + PO4-3 |
12.3 |
|
|
Bisulfide ion |
HS- = H+ + S-2 |
13.9 |
|